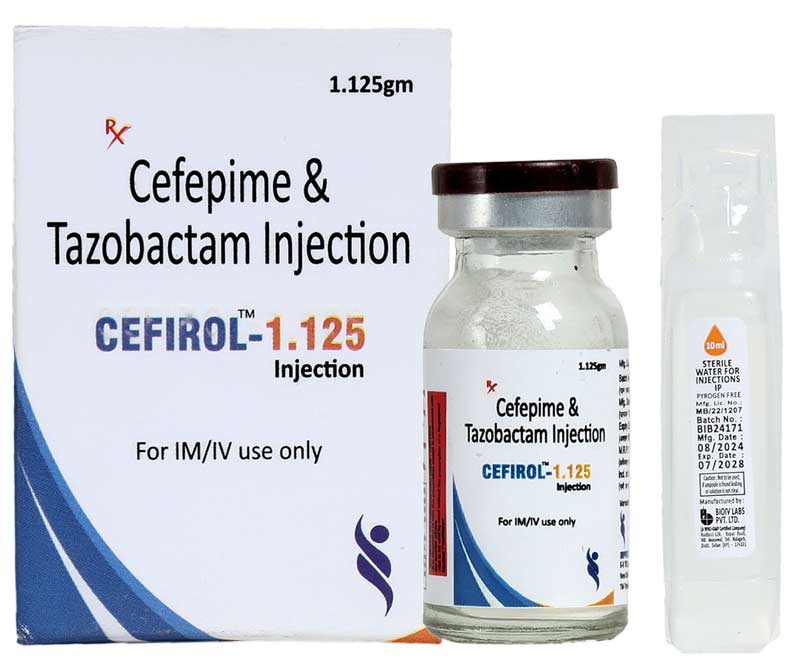
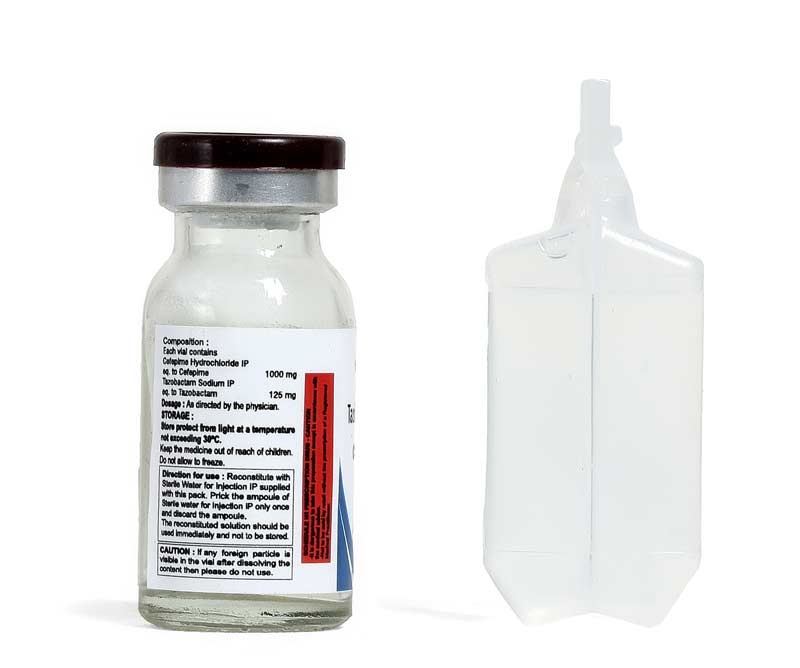
Cefirol-1.125 Injection
Cefirol-1.125 Injection is a broad-spectrum antibacterial combination of Cefepime, a fourth-generation cephalosporin, and Tazobactam, a beta-lactamase inhibitor. This formulation is designed to effectively treat moderate to severe bacterial infections, including those caused by beta-lactamase-producing and resistant organisms.
Cefepime acts by inhibiting bacterial cell wall synthesis, while Tazobactam protects Cefepime from enzymatic degradation, enhancing its antibacterial activity. Together, they provide extended coverage against a wide range of gram-positive and gram-negative pathogens, making Cefirol-1.125 suitable for hospital-based infection management.
It is commonly indicated for respiratory tract infections, urinary tract infections, skin and soft tissue infections, intra-abdominal infections, septicemia, and other complicated infections. The injection is intended for IM/IV administration only under medical supervision.
Manufactured under stringent quality standards, Cefirol-1.125 Injection ensures high purity, stability, and consistent therapeutic performance, making it a dependable choice for healthcare professionals.
Key Features:
- Cefepime & Tazobactam Injection 1.125 g
- Broad-spectrum antibacterial coverage
- Effective against resistant bacterial strains
- For IM/IV use only
- Suitable for hospital and critical care settings
Cefirol-1.125 Injection supports effective infection control and improved patient outcomes through reliable antibiotic therapy.
Disclaimer:
This information is for registered medical practitioner only. Anyone other than medical practitioner should consult medical practitioner before using this product.